Battery Design for Patch Cardiac Monitors
Practical Engineering Guidance for Long-Term Wearable ECG Devices
Medical Battery / ECG Patch / Custom Lithium Battery
Key Requirements for Patch Cardiac Monitors
Patch cardiac monitors (adhesive ECG patches) have become a standard solution for ambulatory rhythm detection, particularly in long-term monitoring scenarios such as arrhythmia detection. These devices are typically required to operate continuously for 7–14 days without interruption.
This requirement creates a highly constrained power budget. Multiple subsystems—including sensors, low-noise analog front ends, microcontrollers, Bluetooth (BLE) or proprietary radios, and onboard data storage—consume power simultaneously. Even though each module may operate at low current individually, their combined load over long durations makes power optimization critical.
In addition to electrical performance, the battery must meet strict mechanical constraints. The device must remain lightweight, ultra-thin, and comfortable for skin attachment, while also maintaining sufficient energy capacity. This is why many product teams ultimately adopt
custom lithium battery solutions, where the cell geometry and discharge profile are optimized specifically for the device architecture.
Another often underestimated requirement is user comfort and skin compatibility. Battery thickness, edge design, and thermal characteristics directly affect patient compliance. Even small increases in thickness or temperature rise can reduce wearability.
Recommended Battery Parameters for Patch Cardiac Monitors
For patch cardiac monitors, battery selection must balance continuous operating time, patient comfort, device thickness, and medical safety requirements. The table below summarizes typical design parameters engineers may use as a reference during early-stage product development and battery customization.
| Parameter | Recommended Range / Target | Engineering Notes |
|---|---|---|
| Battery Chemistry | Li-ion pouch / Lithium polymer / Primary lithium coin cell | Selection depends on rechargeability, device thickness, and required operating life. |
| Nominal Voltage | 3.0V–3.7V | Common range for wearable ECG electronics, BLE modules, and low-power MCUs. |
| Typical Capacity | 100mAh–600mAh | Capacity depends on average current consumption and target wear duration of 7–14 days. |
| Operating Time | 7–14 days continuous monitoring | Should be validated with real-world power profiling, not only theoretical calculations. |
| Average Current Consumption | 50μA–1mA | Varies based on sensing frequency, BLE transmission interval, memory writes, and firmware strategy. |
| Maximum Thickness | Typically 3mm–8mm battery section | Thin-profile design is critical for comfort, adhesion, and long-term skin wearability. |
| Cell Shape | Ultra-thin rectangular / curved custom pouch | Custom geometry helps fit compact adhesive patch housings. |
| Peak Discharge Capability | Designed for short wireless burst loads | Battery must support BLE transmission, flash writes, and transient current spikes. |
| Protection Features | Overcharge, over-discharge, overcurrent, short-circuit protection | Essential for patient safety and regulatory compliance. |
| Operating Temperature | Customized to product environment, often near body temperature range | Thermal stability is important because the device remains attached to skin for extended periods. |
| Cycle / Use Model | Single-use or rechargeable depending product design | Disposable patches may prioritize primary lithium; reusable monitors may use rechargeable lithium polymer cells. |
| Compliance Requirements | IEC 62133, UL 1642, UN38.3 | Testing scope should align with medical device certification and transport requirements. |
| Recommended Battery Type | Custom lithium polymer battery | Best suited for thin, lightweight, wearable patch monitor designs requiring shape flexibility. |
Choosing the Right Chemistry and Form Factor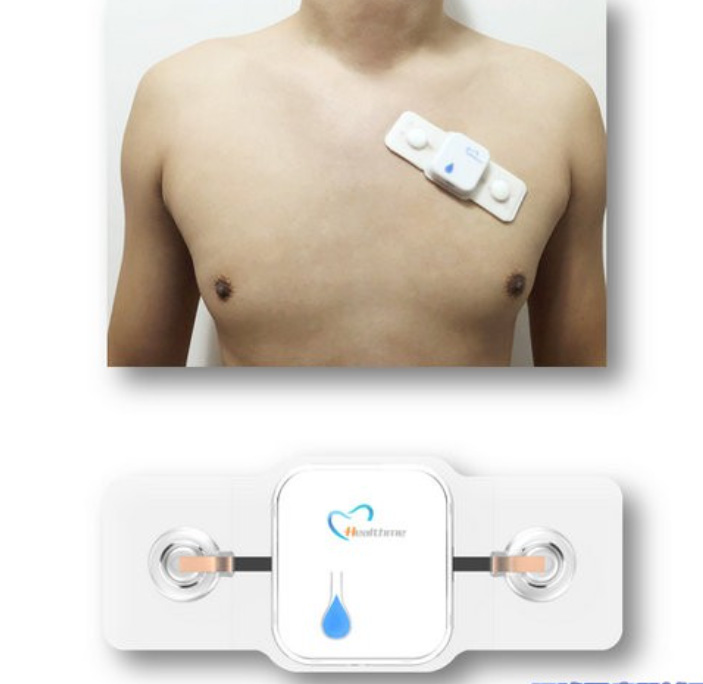
For patch cardiac monitors, both rechargeable and primary lithium battery chemistries are used depending on the design strategy and product lifecycle.
- Li-ion pouch cells: These provide high energy density and flexible form factors, making them ideal for thin wearable designs. Their laminated structure allows customization in shape and size.
- Coin cells (Li-MnO₂ / Li-SoCl₂): These are suitable for ultra-low-power applications where rechargeability is not required. They offer long shelf life and stable discharge but limited peak current capability.
- Custom lithium polymer batteries: These are engineered solutions tailored to specific device requirements, including thickness, curvature, and capacity distribution.
Energy density is one of the most critical parameters, as it directly impacts device size and operating duration. Typical lithium-ion systems provide approximately 150–300 Wh/kg depending on materials and packaging.
However, higher energy density often introduces trade-offs, such as increased thermal management requirements and higher material costs. Engineers must balance these factors based on the target use case.
In practice, custom lithium polymer batteries are widely adopted because they allow designers to precisely match the battery to the mechanical envelope of the patch device.
Designing for Safety and Regulatory Compliance
Safety is a non-negotiable requirement in medical battery design. Patch monitors are worn directly on the skin for extended periods, which increases the importance of thermal stability and leakage prevention.
Common regulatory standards include:
- IEC 62133 (battery safety)
- UL 1642 (cell-level safety)
- FDA-recognized consensus standards
A robust battery system design typically includes:
- Battery Management System (BMS) or protection IC
- Overcurrent and short-circuit protection
- Undervoltage cutoff
- Thermal protection mechanisms
- Mechanical reinforcement and sealing
These features reduce the risk of thermal runaway, electrolyte leakage, or unintended activation. For regulated healthcare environments, it is recommended to work with specialized
medical battery solution providers.
Custom Pack Engineering: Trade-offs and Design Variables
Designing a custom battery pack for a patch cardiac monitor involves multiple trade-offs:
- Capacity vs. Thickness: Increasing capacity improves runtime but directly impacts device thickness and flexibility.
- Peak Current Capability: Wireless communication and flash memory writes introduce short bursts of high current demand.
- Manufacturing Constraints: Automated assembly, tab welding (often ultrasonic), and coating processes must be considered early in design.
- Environmental Performance: Batteries must operate reliably under varying temperature and humidity conditions.
In real-world projects, these variables are iteratively optimized through prototyping and testing. Engineers often collaborate closely with suppliers providing
custom battery packs to reach the optimal balance.
Integrating Battery Electronics and Firmware
Hardware design alone is not sufficient. Firmware plays a critical role in extending battery life and ensuring predictable performance.
Effective power management strategies include:
- Duty cycling of sensors and communication modules
- Event-driven data acquisition instead of continuous high-rate sampling
- Low-power BLE communication modes
- Deep sleep states for microcontrollers
In addition, battery monitoring functions should be implemented to track:
- State of Charge (SOC)
- End of Life (EOL)
- Voltage and temperature conditions
These parameters are critical for clinical reliability and remote diagnostics.
Estimating Battery Life: A Practical Model
Engineers often use a simplified model to estimate device runtime:
Lifetime (days) = Capacity (mAh) / Average Current (mA) / 24
While this formula provides a useful baseline, real-world performance depends on dynamic load profiles, temperature, and battery aging.
For example, intermittent BLE transmission and flash writes can significantly impact average current consumption, even if idle current remains low.
Therefore, accurate lifetime estimation requires both theoretical calculation and empirical validation through power profiling.
Production and Supply Chain Considerations
Selecting the right battery supplier is critical for product success. Key evaluation factors include:
- Experience with medical-grade battery systems
- Ability to meet custom specifications
- Quality control and traceability
- Availability of certification documentation (IEC, UN38.3)
Working with a qualified supplier reduces development risk and accelerates time-to-market.
Battery Testing and Validation
Comprehensive validation is essential for medical devices. Testing should include:
- Cycle life testing
- Storage and aging tests
- Mechanical shock and vibration testing
- Temperature cycling
- Biocompatibility testing for skin-contact materials
Third-party accredited laboratories are typically used for UL and IEC certification to support regulatory submissions.
Conclusion
Designing a battery system for patch cardiac monitors requires a multidisciplinary approach that combines electrochemistry, mechanical design, firmware optimization, and regulatory compliance.
By carefully balancing energy density, safety, manufacturability, and user comfort, engineers can deliver reliable and clinically effective wearable devices.
If you are working on a medical wearable project, you can explore
custom battery solutions or
contact our engineering team for support.


