Custom Battery Design for Handheld Ultrasound Devices
Engineering Reliable Power for Portable Medical Imaging Systems
Medical Device Battery
To meet these requirements, many medical device manufacturers work with specialized suppliers to design custom lithium polymer battery packs. These solutions optimize energy density, thermal stability, and electrical performance while ensuring compliance with medical safety standards. A well-designed battery architecture not only improves device reliability but also enhances the user experience by providing longer runtime and predictable charging cycles.
Why Power Design for HHUS Devices Is Challenging
Power system design for handheld ultrasound devices involves multiple engineering challenges that go beyond typical portable electronics. The first challenge is the highly dynamic power demand generated by ultrasound imaging electronics. During scanning, the transducer array and signal processing hardware require short bursts of high current, especially when operating advanced imaging modes such as Doppler or harmonic imaging.
If the battery pack cannot respond quickly to these transient loads, voltage drops may occur that interrupt imaging or cause system instability. For this reason, battery cells used in ultrasound systems must exhibit low internal resistance and excellent pulse current capability. High-performance lithium polymer cells are commonly selected because they can provide both high energy density and strong current output characteristics.
Another challenge is thermal management. Handheld ultrasound devices often feature sealed housings to prevent fluid ingress and simplify disinfection procedures. These designs typically aim for IPX7 waterproof ratings, which restrict airflow inside the device. Engineers must therefore carefully manage heat distribution between the ultrasound electronics and the battery pack to prevent overheating.
Three Proven Battery Architectures for Portable Ultrasound
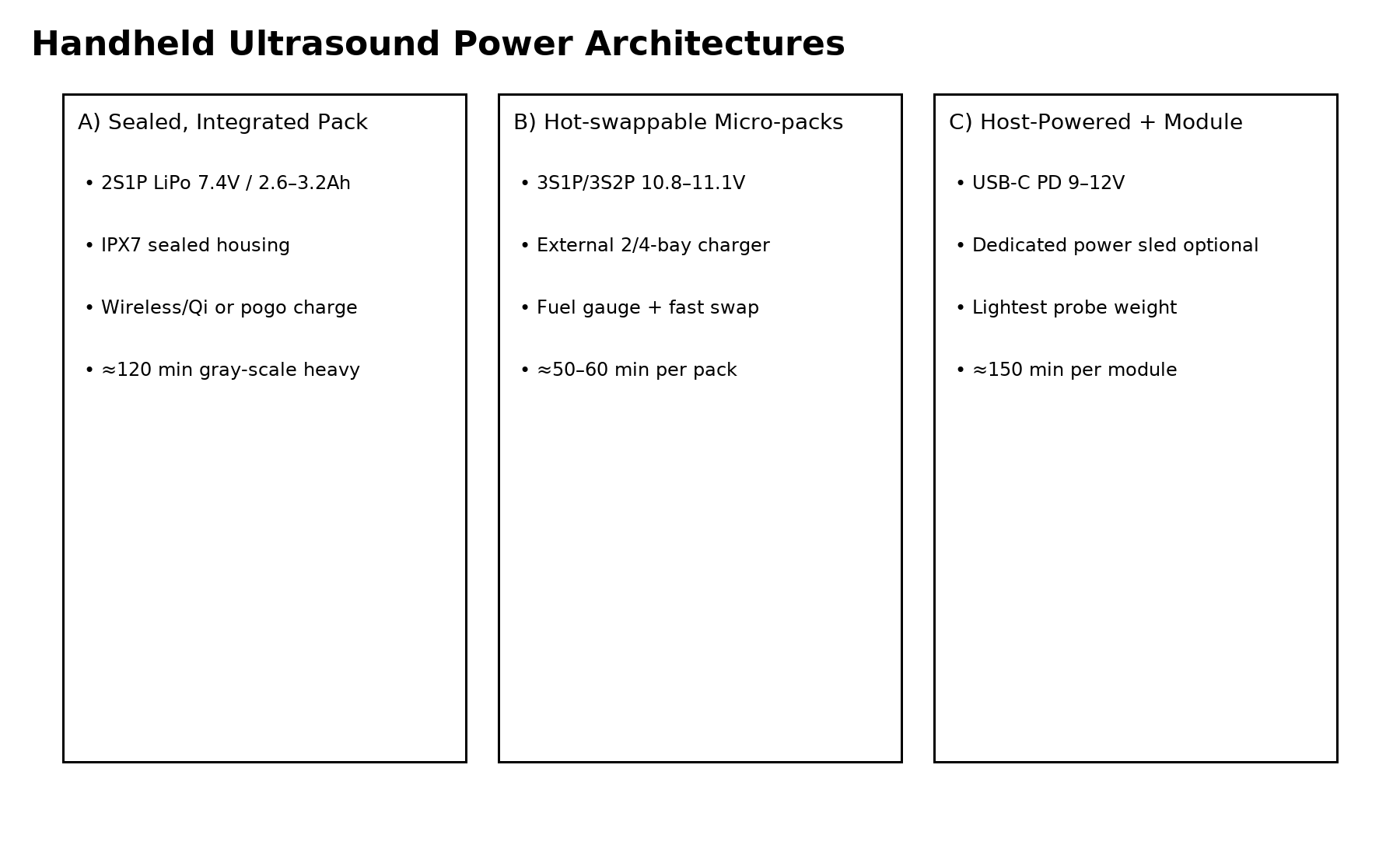
Integrated Sealed Battery Pack
The most compact handheld ultrasound devices use fully integrated battery packs sealed inside the probe housing. This design minimizes device weight and simplifies the user experience, as clinicians do not need to remove or replace batteries during operation. Typical packs use a 2S lithium polymer configuration delivering around 7.4 V and 2.6–3.2 Ah capacity.
Charging is usually performed through wireless charging pads or magnetic docking connectors. This architecture provides excellent hygiene control because the device can be easily wiped down without exposed electrical contacts. However, thermal management becomes more complex because both the electronics and battery share the same enclosure.
Hot-Swappable Micro Battery Modules
Hospitals that require continuous scanning capability often prefer removable battery modules. These packs typically use cylindrical lithium cells such as 18650 or 21700 in 3S1P or 3S2P configurations. Each pack can provide approximately 50 to 60 minutes of scanning time and can be replaced within seconds when depleted.
Hot-swappable designs allow clinicians to maintain uninterrupted operation by swapping packs while another battery is charging in a docking station. This architecture is frequently used in emergency departments and mobile healthcare environments. Battery packs designed for these systems often incorporate advanced fuel gauge chips and protective circuits.
Host-Powered Hybrid Systems
Some handheld ultrasound probes rely on external devices such as tablets or smartphones for both computing and power supply. In this architecture, the probe itself remains extremely lightweight while the energy source is provided through a USB-C Power Delivery interface or external battery module.
Hybrid systems can achieve longer operating times while keeping the probe small and ergonomic. However, they introduce additional complexity in cable management and charging logistics.
Battery Chemistry and Pack Configuration
Lithium-ion and lithium polymer batteries dominate the portable medical device market due to their excellent balance between energy density, weight, and performance. Most handheld ultrasound devices use NMC-based lithium polymer cells because they provide high capacity while maintaining compact form factors.
In applications where safety and cycle life are prioritized over energy density, LiFePO4 battery technology may be considered. LiFePO4 cells offer improved thermal stability and longer cycle life, making them suitable for rugged medical equipment or industrial diagnostic devices.
Typical handheld ultrasound battery packs operate at either 7.4 V (2-series configuration) or 11.1 V (3-series configuration). The optimal voltage platform depends on the internal power architecture of the device and the efficiency of the DC-DC converters used to drive the ultrasound transducer circuits.
Battery Management System and Telemetry
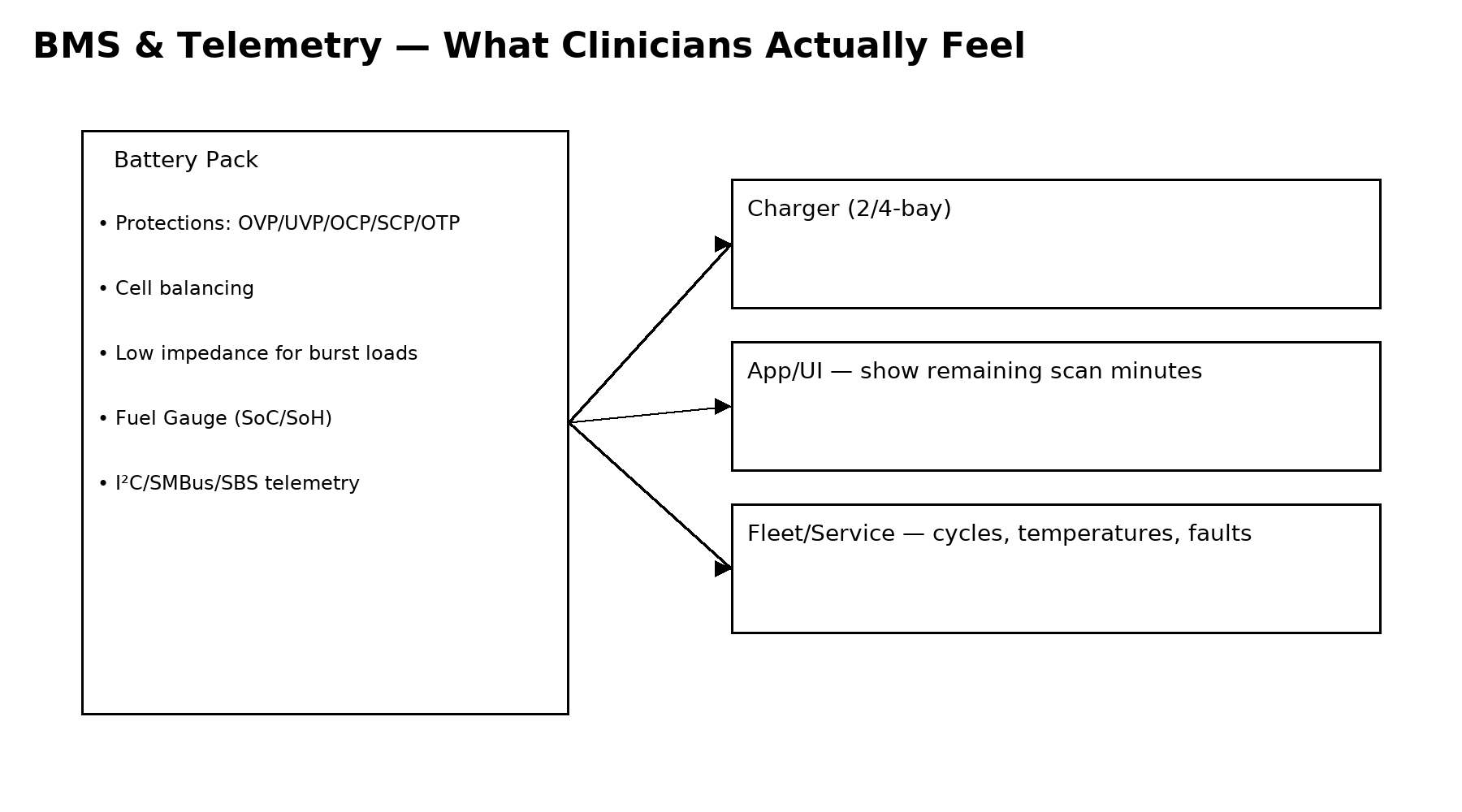
A sophisticated Battery Management System (BMS) ensures safe and reliable operation of lithium battery packs in medical devices. The BMS monitors cell voltages, temperature, current flow, and charging status while protecting the battery from abnormal conditions such as over-charge, over-discharge, and short circuits.
Modern battery packs often integrate fuel gauge technology that estimates the battery’s state of charge and remaining runtime. These measurements can be communicated to the ultrasound system through SMBus or I²C interfaces, allowing clinicians to monitor battery levels directly from the device display.
Advanced telemetry features also enable healthcare providers to track battery usage data across multiple devices. By recording cycle counts and temperature events, maintenance teams can schedule proactive battery replacement before performance degrades.
Charging Methods for Medical Ultrasound Devices
Several charging approaches are used in handheld ultrasound systems depending on device architecture. Multi-bay chargers are commonly used for removable battery packs, allowing hospitals to charge several batteries simultaneously while maintaining individual monitoring for each slot.
Wireless charging systems are popular for sealed probes because they eliminate exposed connectors and simplify infection control procedures. However, engineers must carefully manage heat buildup associated with inductive charging technologies.
Magnetic pogo-pin docking stations provide another practical solution. These systems combine efficient electrical connections with simple mechanical alignment and easy cleaning.
Environmental and Durability Engineering
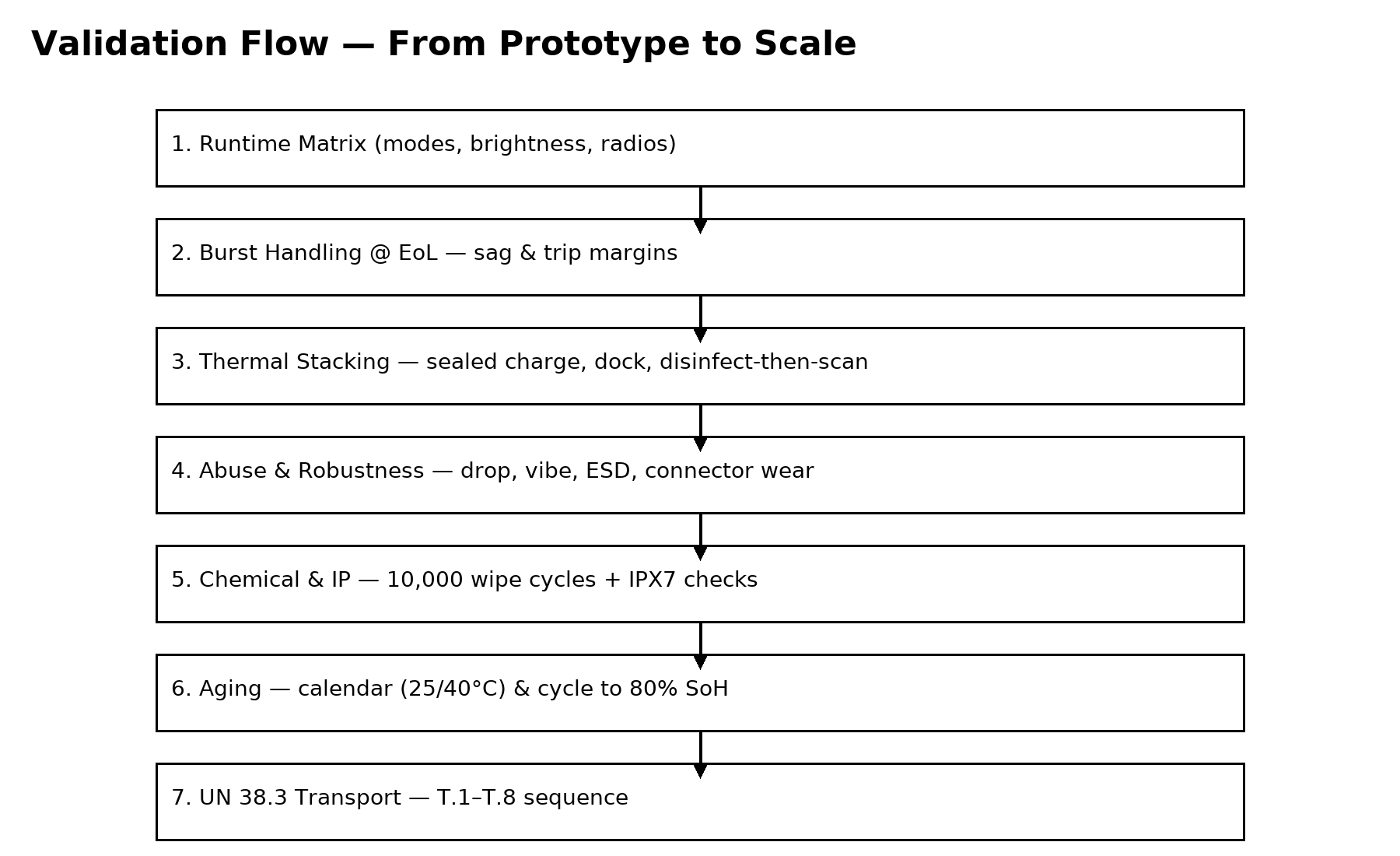
Medical environments expose devices to frequent cleaning cycles and demanding operating conditions. Handheld ultrasound systems are often disinfected multiple times per day using alcohol or bleach-based solutions. Battery pack materials, adhesives, and labels must therefore be tested for chemical compatibility to prevent degradation.
Engineers also conduct mechanical durability testing to ensure that battery packs remain safe after drops, vibration, and repeated connector insertions. These validation procedures help ensure reliable operation throughout the device’s lifecycle.
Medical Safety and Compliance Standards
Battery packs used in medical equipment must meet strict international safety requirements. Common standards include IEC 62133-2 for lithium battery safety and UL 2054 certification for battery packs. In addition, handheld ultrasound systems must comply with IEC 60601-1 medical electrical safety standards and related electromagnetic compatibility regulations.
For global transportation, lithium batteries must pass the UN 38.3 test sequence before being shipped by air or sea. Manufacturers must also provide documentation such as lithium battery test summaries and material safety data sheets.
Example Battery Configuration for Handheld Ultrasound
| Application | Battery Configuration | Nominal Voltage | Capacity | Runtime |
|---|---|---|---|---|
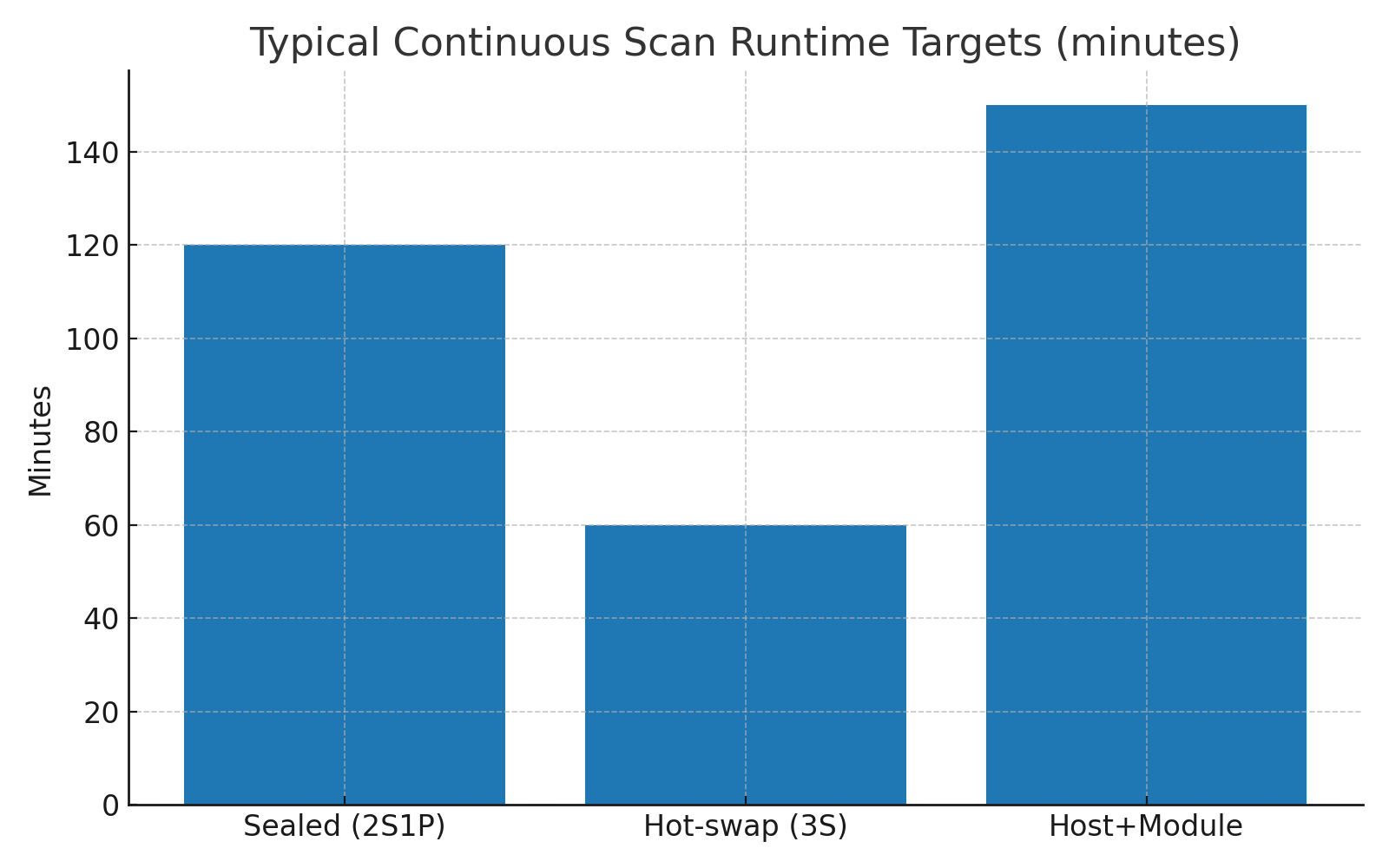
| Ultra-portable probe | 2S1P LiPo | 7.4V | 2.6–3.2Ah | ~120 minutes |
| Hot-swap clinical device | 3S1P 18650 | 10.8V | 2.5–3Ah | ~60 minutes per pack |
| Tablet-connected probe | External module | 9–12V | Custom | ~150 minutes |
Companies developing portable medical devices often collaborate with experienced battery manufacturers to design custom packs optimized for their product architecture. For example, PKNERGY provides specialized battery development services for medical electronics as part of its medical device power solution.
Frequently Asked Questions
What type of battery is used in handheld ultrasound devices?
Most handheld ultrasound scanners use lithium-ion or lithium-polymer batteries due to their high energy density and ability to deliver stable power during imaging.
How long does a handheld ultrasound battery last?
Depending on battery capacity and imaging mode, typical runtime ranges from 50 minutes to more than two hours of continuous scanning.
Can handheld ultrasound devices use removable batteries?
Yes. Some devices use sealed integrated batteries, while others rely on hot-swappable battery packs to support continuous clinical operation.
Custom Lithium Polymer Battery
Medical Device Battery Solution
Get a Quote


